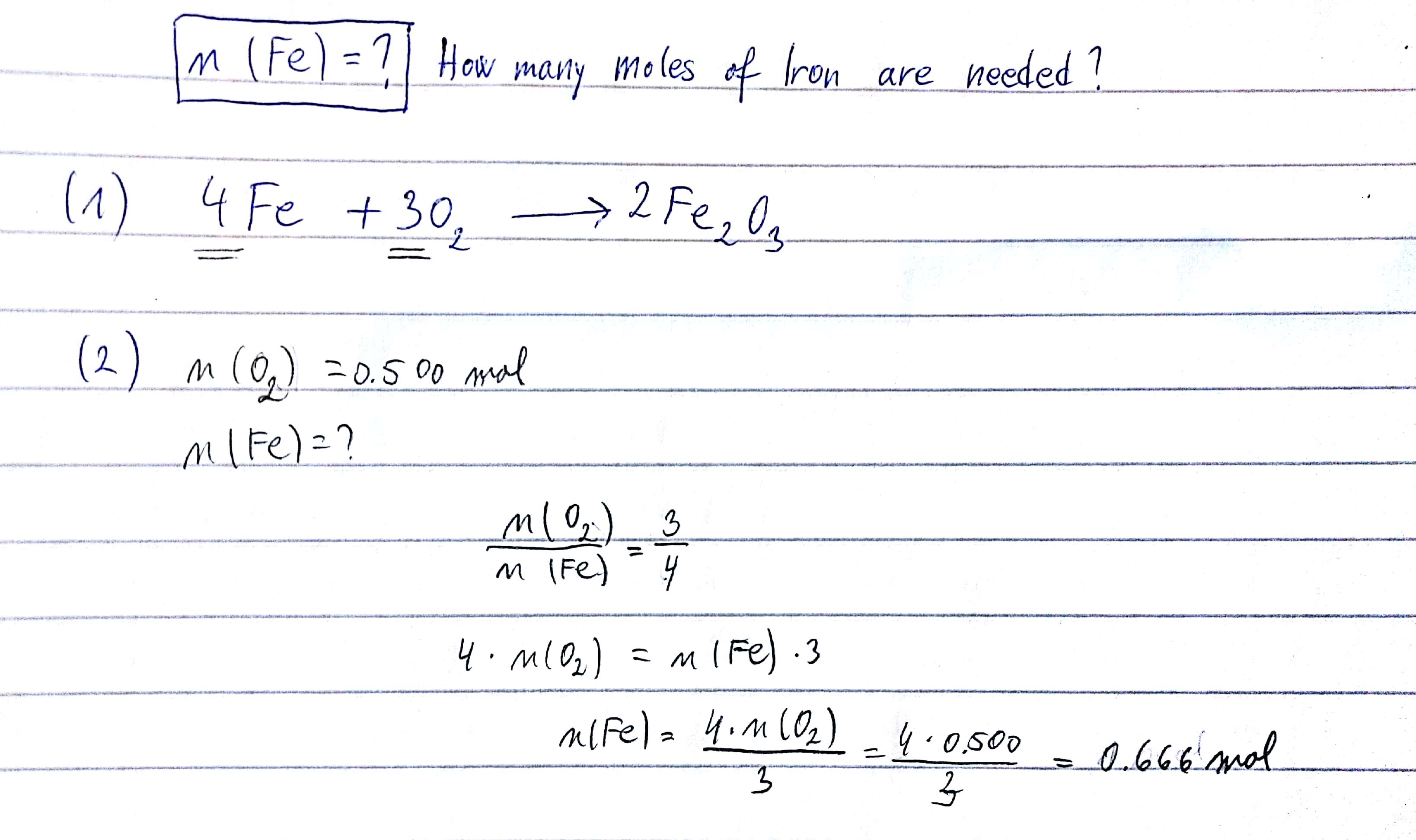XRF pattern of ocher particles: 1-FeO(OH) goethite; 2-Fe2O3 hematite;... | Download Scientific Diagram
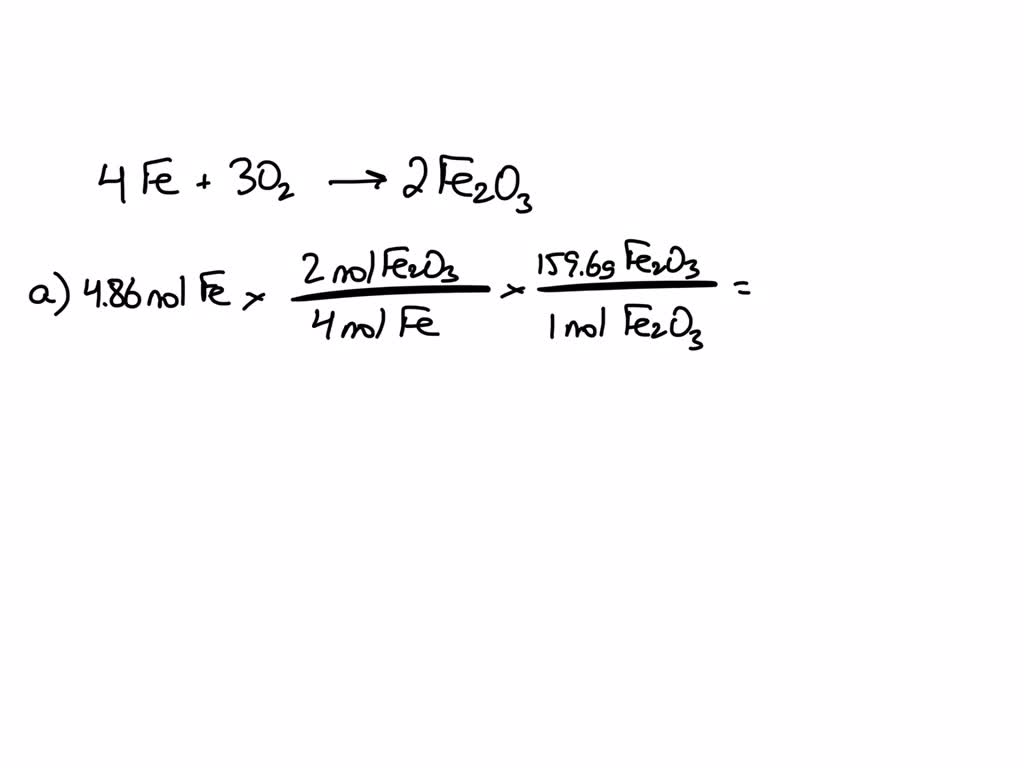
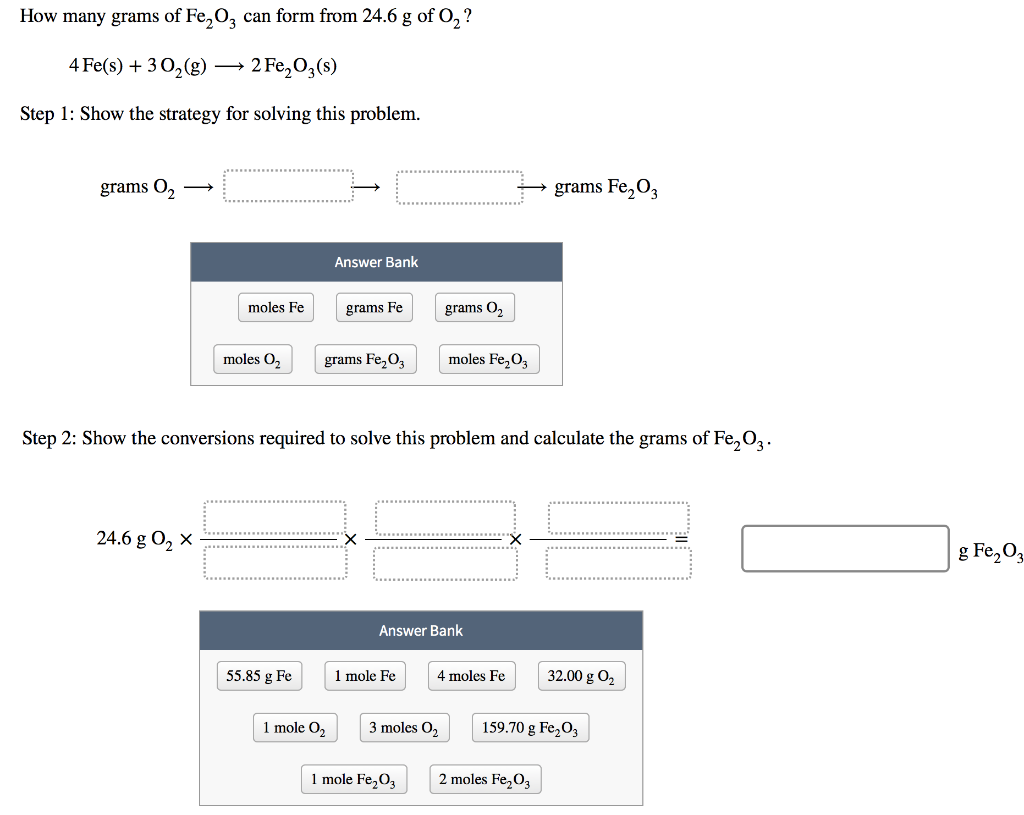
SOLVED: Given the reaction 4Fe + 3O2= 2Fe2O3 a. How many grams of Fe2O3 will be formed from 4.86 moles Fe reacting with sufficient oxygen gas? b. How many grams of Fe

SOLVED: Please provide solution so I can review. Using the reaction: 4Fe + 3O2 → 2Fe2O3 answer the following. How many moles of Fe2O3 are produced when 0.275 moles of Fe is