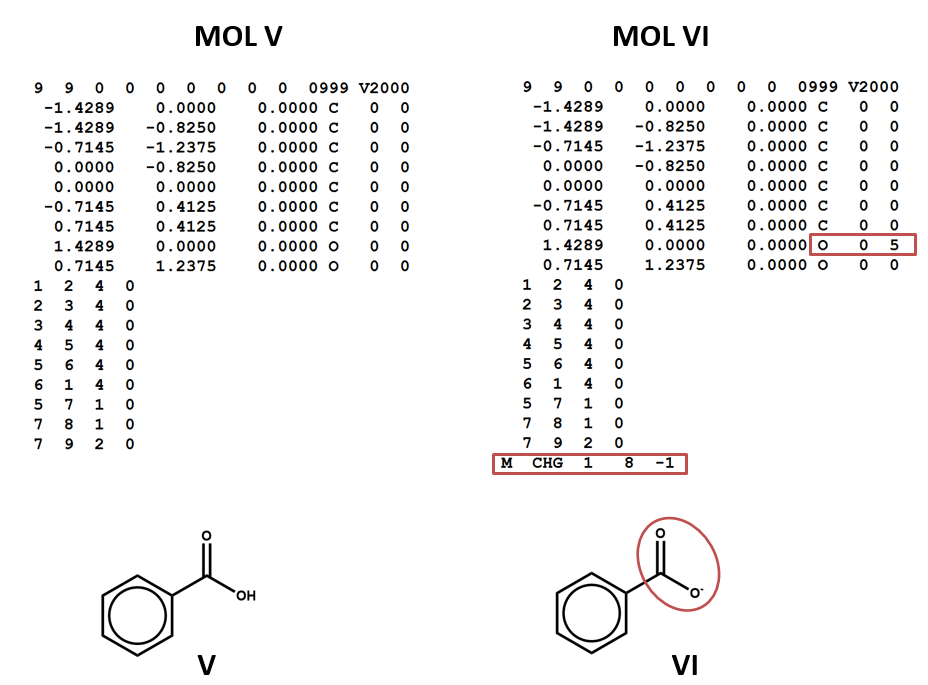
SOLVED: A compound of carbon and hydrogen contains 92.3% C and has a molar mass of 78.1 g/mol. Select all that is correct. A. The empirical formula is CH. B. The molecular

Hydrated Ions: From Individual Ions to Ion Pairs to Ion Clusters | The Journal of Physical Chemistry B









:max_bytes(150000):strip_icc()/vitamin-c-molecular-model-483948223-582c8a523df78c6f6a473f1c.jpg)