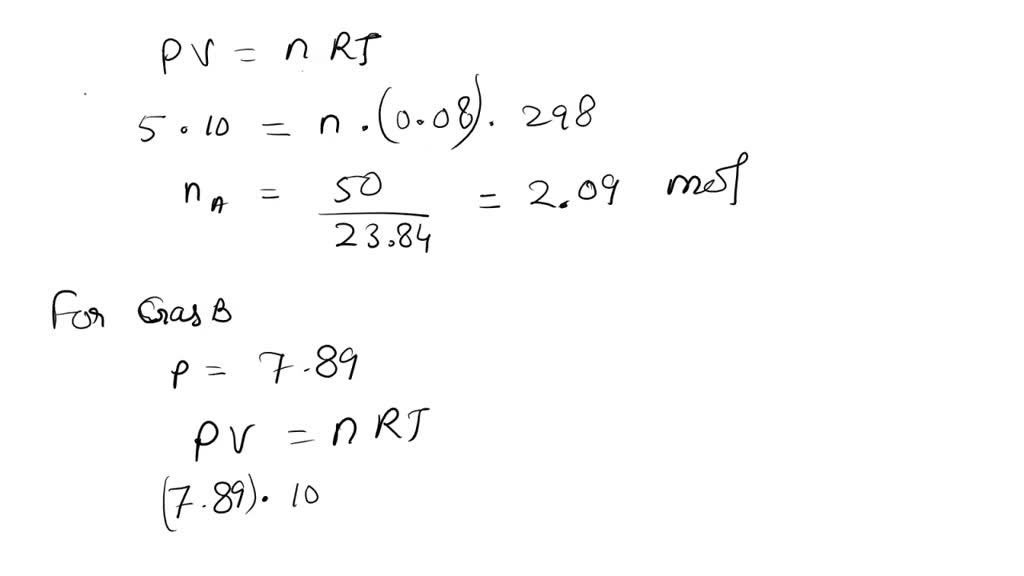
SOLVED: At 25.0° C, a 10.00 L vessel is filled with 5.00 atm of Gas A and 7.89 atm of Gas B. What is the mole fraction of Gas B?

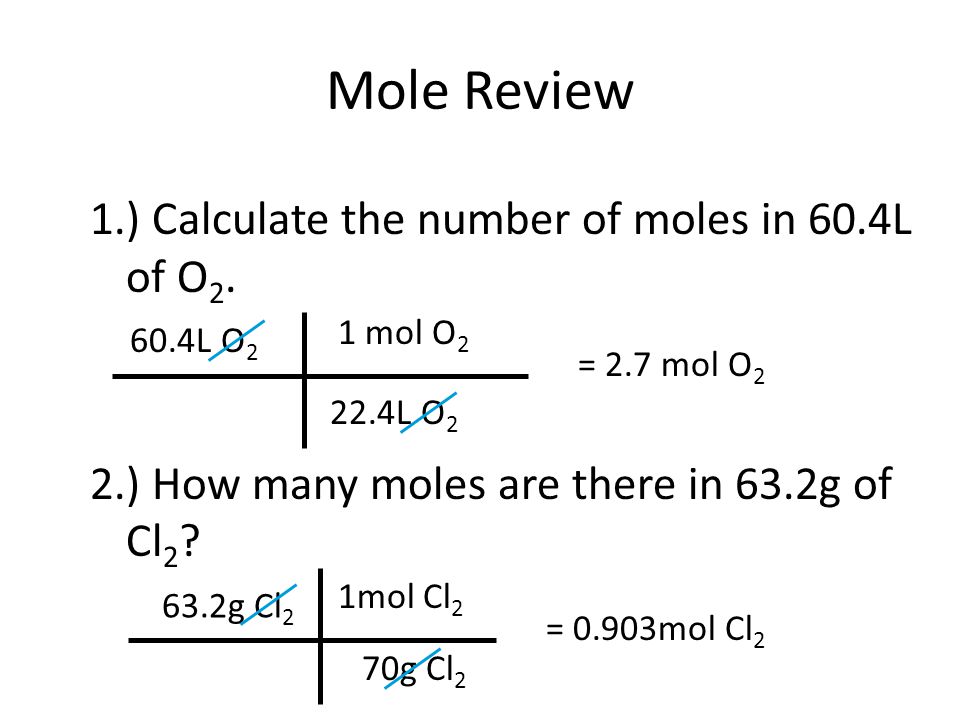
SOLVED:Use molar volume to calculate each of the following at STP: a. the number of moles of O2 in 44.8 L of O2 gas b. the volume, in liters, occupied by 0.420

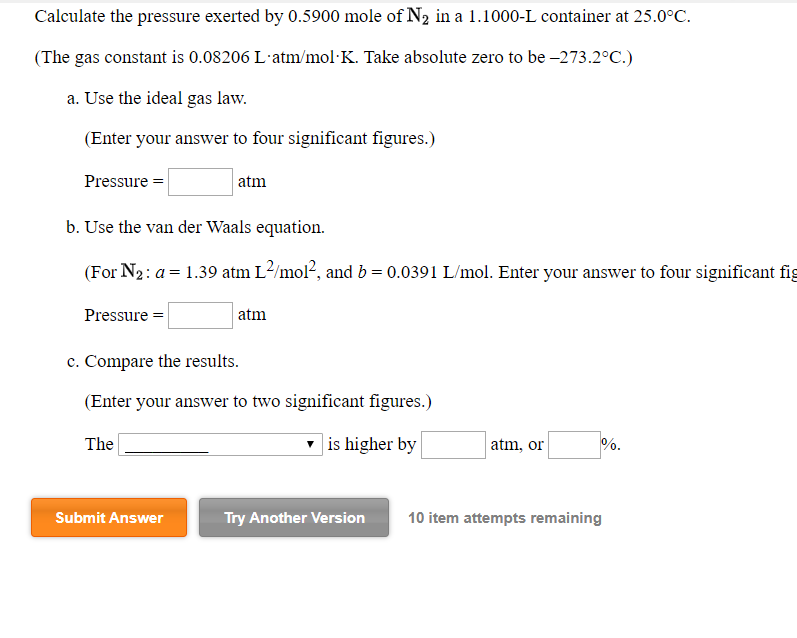
Calculate the temperature of 2 moles of sulphur dioxide gas contained in a 5 L vessel at 10 bar pressure. Given that for SO(2) gas, van der Waals constants are : a=6.7

SOLVED:Use molar volume to calculate each of the following at STP: a. the number of grams of Ne contained in 11.2 L of Ne gas b. the volume, in liters, occupied by









![ANSWERED] 1 mol = 22.4 L 1 mol = 6.02 x 1023 repres... - Physical Chemistry ANSWERED] 1 mol = 22.4 L 1 mol = 6.02 x 1023 repres... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/80755040-1659993191.1665888.jpeg)


