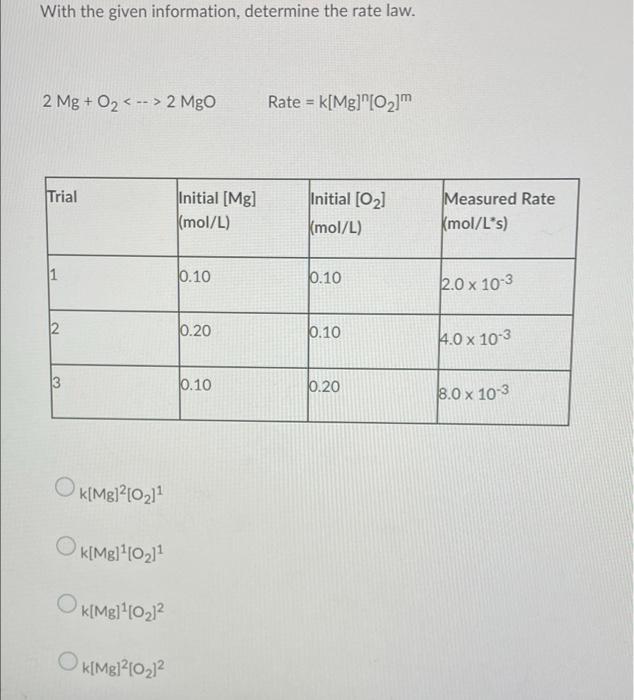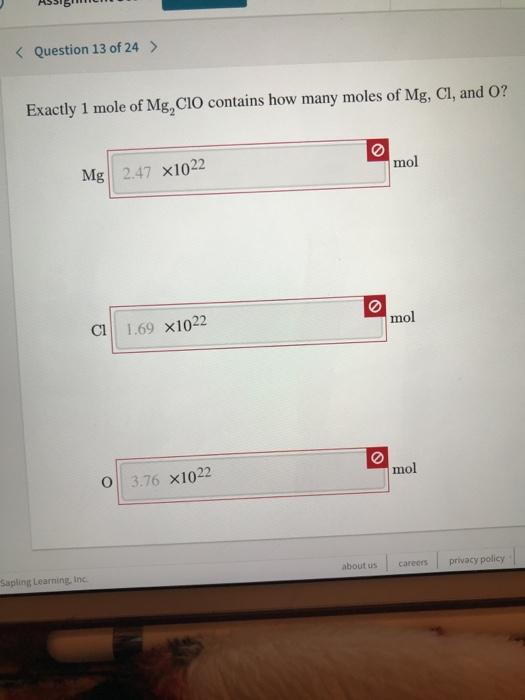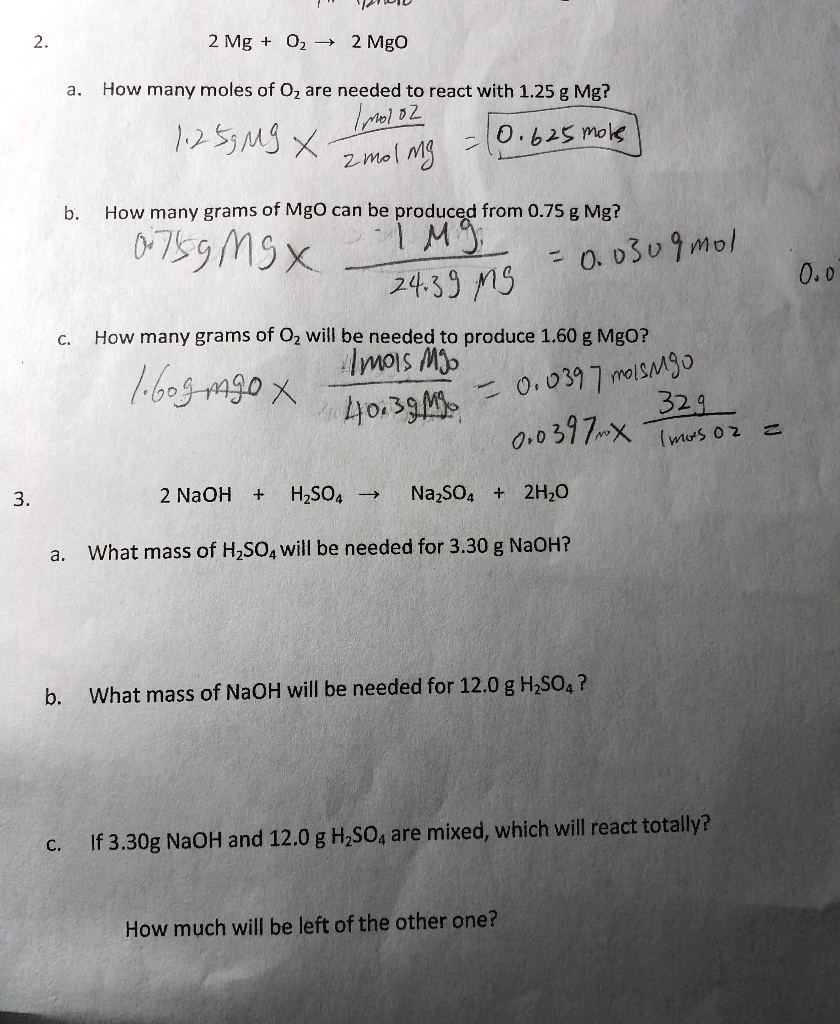
SOLVED: 2 Mg 0z 7 2 Mgo How many moles of Oz are needed to react with 1.25 g Mg? Mol 22 12S5MS 0.62E mok 2m( mS How many grams of MgO
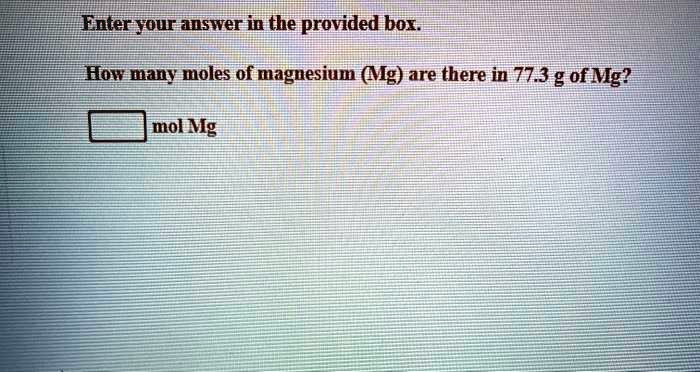
SOLVED: Enter your answer in the provided box How many moles of magnesium ( Mg) are there in 77.3 g of Mg? mol Mg

The Mole Concept. What is a mole? IIn chemistry, a mole is a counting unit. Abbreviated mol. 11 mol = 6.022x10 23 representative particles. Avogadro's. - ppt download

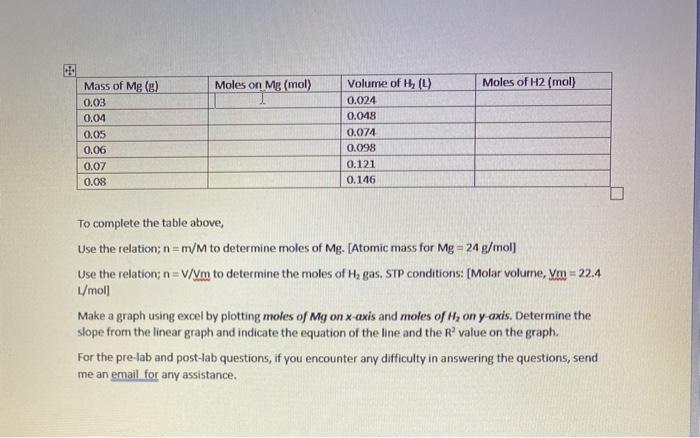
SOLVED: Results (1 pt): Patm (atm) Temperature (K) Volume of Hz produced (L) Moles of Hz produced (mol) Moles of Mg consumed (mol) Experimental atomic mass of Mg (g mol (report to

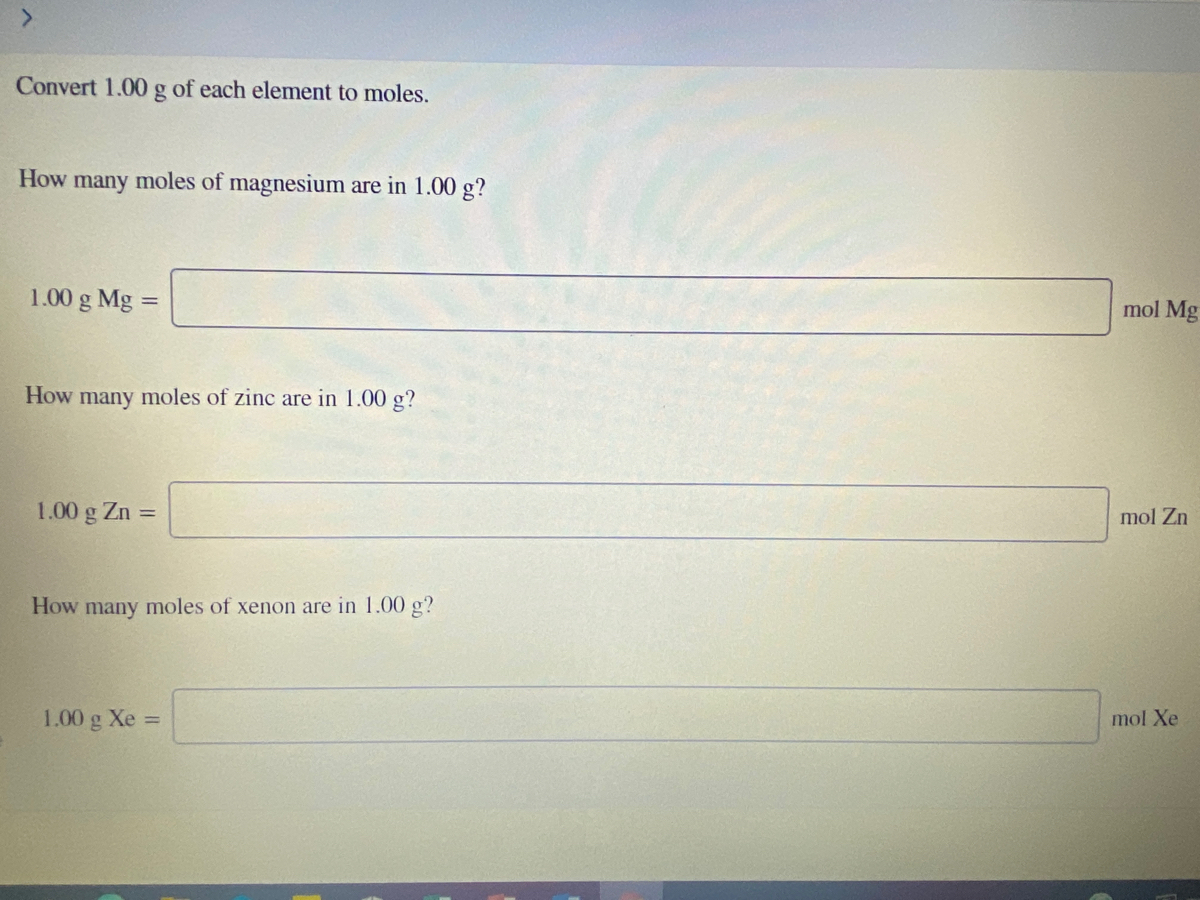





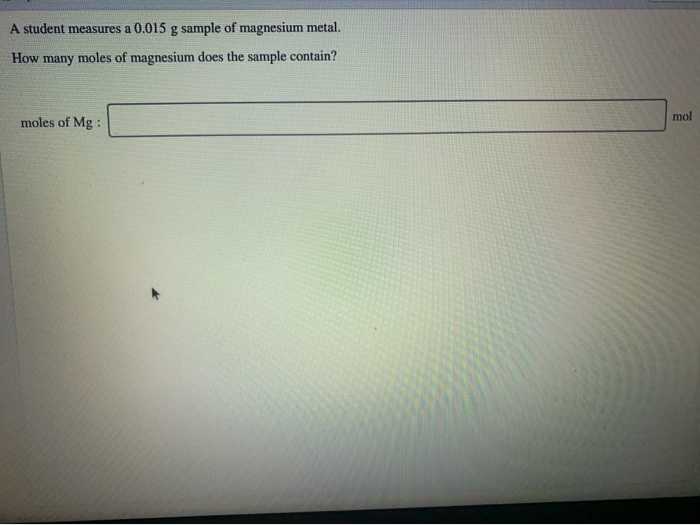
![ANSWERED] How many moles is equivalent to 72.9 g Mg... - Physical Chemistry ANSWERED] How many moles is equivalent to 72.9 g Mg... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/84369751-1657184859.1788638.jpeg)