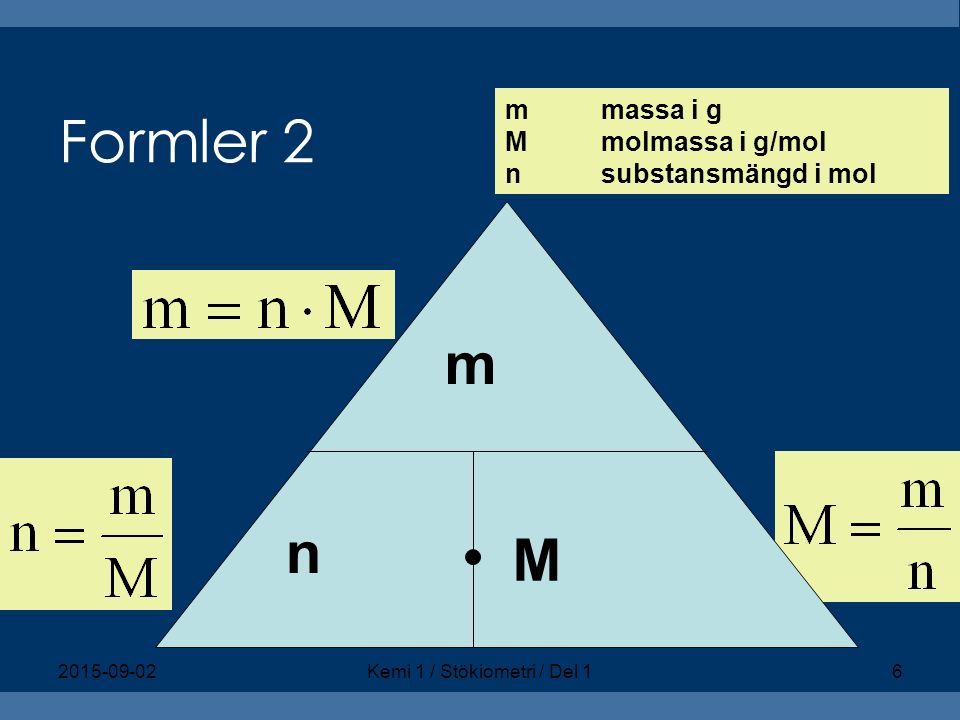
Molformeltriangeln Isolerad På Vitt Förhållandet Mellan Mol Massa Och Molmassa Med Ekvationer-vektorgrafik och fler bilder på Kemi - iStock

MOLAR MASS Molar mass of a substance = mass in grams of one mole of the substance. A compound's molar mass is NUMERICALLY equal to its formula mass. Formula. - ppt download

SOLVED: 12) (5 pts.) A compound has the empirical formula CHzO and molecular weight of 120.10 grams/mol. What is the molecular formula of the compound?
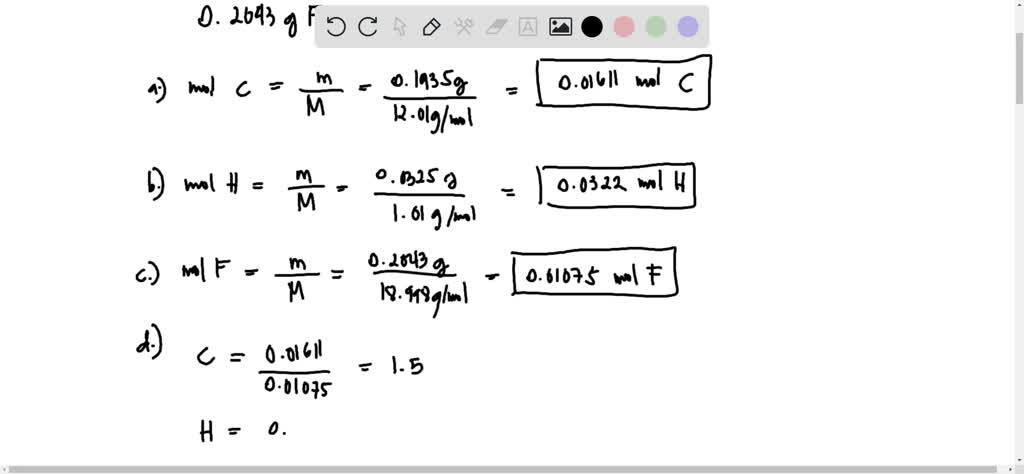
SOLVED: 'An unknown compound is analyzed and found to contain 0.1935 g of carbon, 0.0325 g of hydrogen, and 0.2043 g of fluorine.The molar mass of the compound is 240.23 glmol. a)
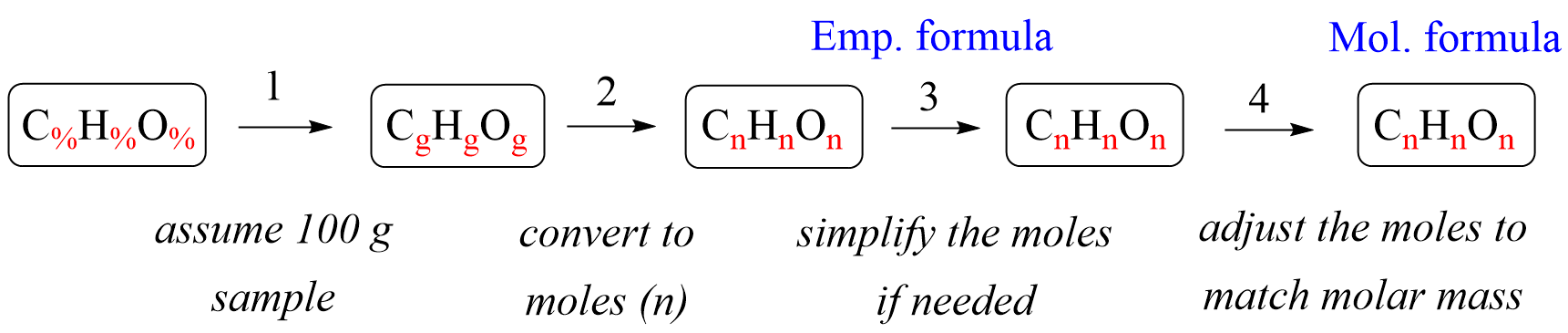
![Molare Masse berechnen • Formel und Berechnung · [mit Video] Molare Masse berechnen • Formel und Berechnung · [mit Video]](https://d1g9li960vagp7.cloudfront.net/wp-content/uploads/2019/10/WP_Molare-Masse_dreieck-1024x576.jpg)