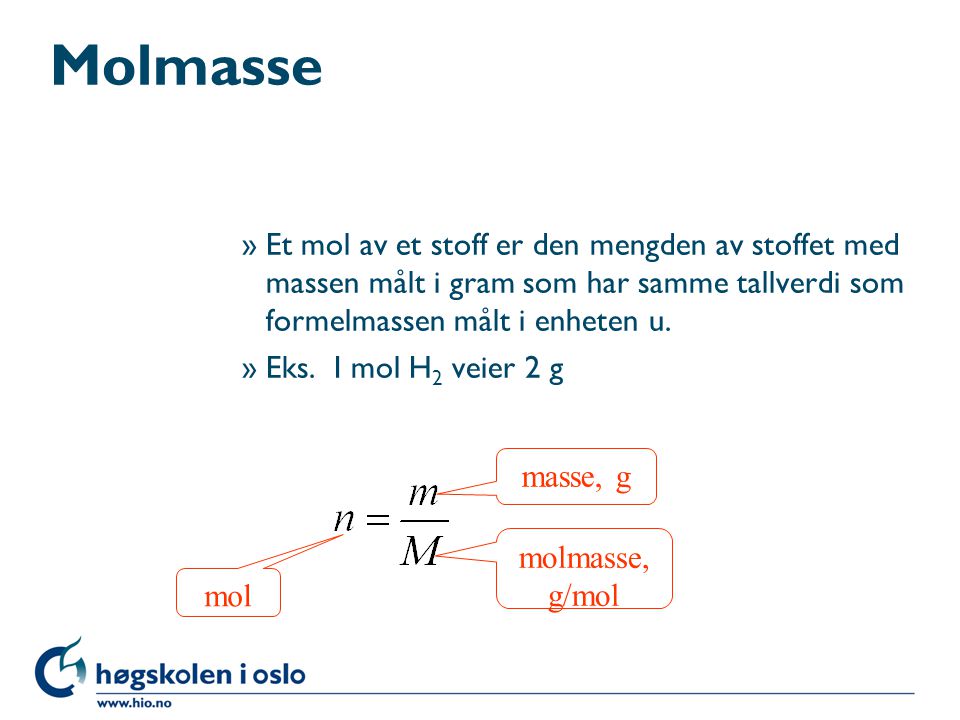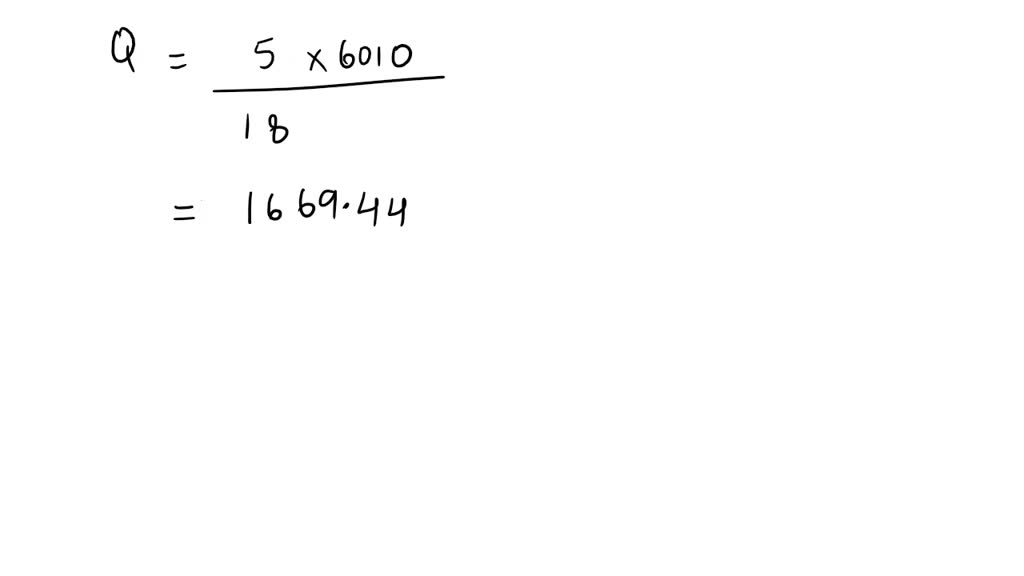
SOLVED: "10.56 How much energy in kilojoules is needed to heat 5.00 g of ice from 10.0 *€ to 30.0 *C? The heat of fusion of water is 6.01 kJ /mol, and
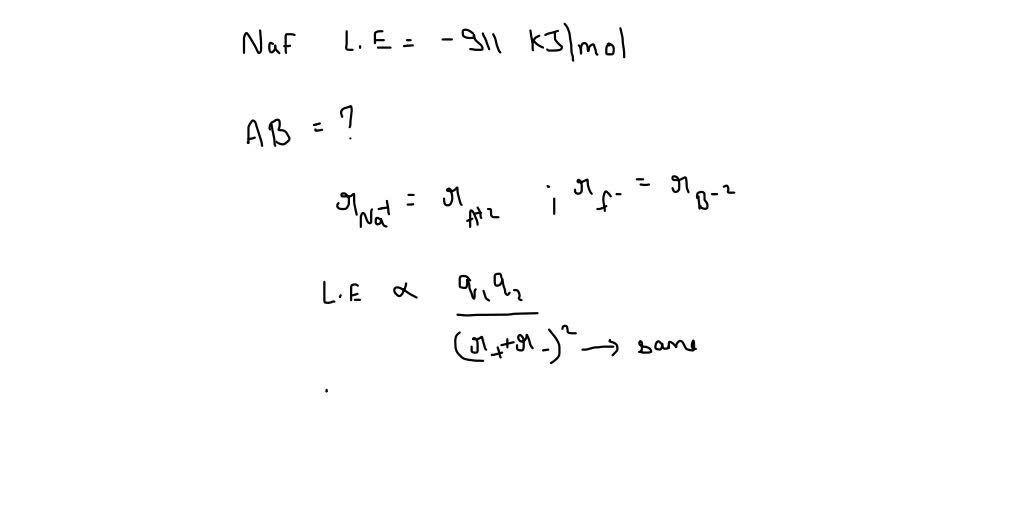
SOLVED: Question 3 of 17 NaF has lattice energy of 911 kIlmol. Consider a generic salt, AB, where A2+ has the same radius as Na+ and B2 has the same radius as





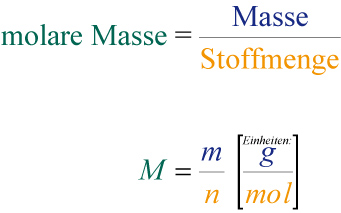
![Molare Masse berechnen • Formel und Berechnung · [mit Video] Molare Masse berechnen • Formel und Berechnung · [mit Video]](https://d1g9li960vagp7.cloudfront.net/wp-content/uploads/2019/10/WP_Molare-Masse_dreieck-1024x576.jpg)
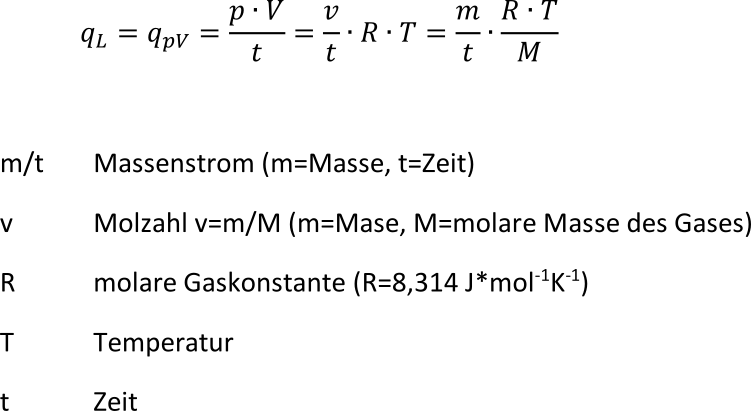
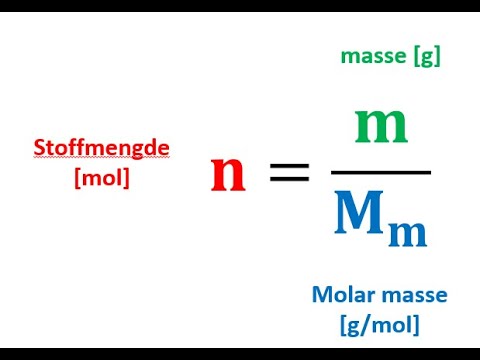

![Mol • einfach erklärt: Berechnung, Umrechnung · [mit Video] Mol • einfach erklärt: Berechnung, Umrechnung · [mit Video]](https://d3f6gjnauy613m.cloudfront.net/system/production/videos/003/617/60e8434046b2a18a42e887d5117b55f87397c228/poster_Thumbnail_Mol.jpg?1639048296)